4-AMINOSULFONYL-1-HYDROXY-2-NAPHTHOIC ACID, 97
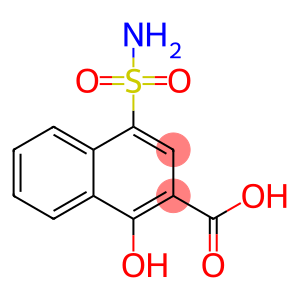
4-Aminosulfonyl-1-hydroxy-2-naphthoic acid
CAS: 64415-15-2
Molecular Formula: C11H9NO5S
4-AMINOSULFONYL-1-HYDROXY-2-NAPHTHOIC ACID, 97 - Names and Identifiers
| Name | 4-Aminosulfonyl-1-hydroxy-2-naphthoic acid |
| Synonyms | 4-Aminosulfonyl-1-hydroxy-2-naphthoic acid 4-Aminosulphonyl-1-hydroxy-2-naphthoic acid 4-aminosulphonyl-1-hydroxy-2-naphthoic acid 1-hydroxy-4-sulfamoylnaphthalene-2-carboxylate 1-hydroxy-4-sulfamoylnaphthalene-2-carboxylic acid 4-AMINOSULFONYL-1-HYDROXY-2-NAPHTHOIC ACID, 97 4-Aminosulfonyl-1-hydroxy-2-naphthalenecarboxylic acid 2-Naphthalenecarboxylic acid, 4-(aminosulfonyl)-1-hydroxy- |
| CAS | 64415-15-2 |
| EINECS | 264-882-0 |
| InChI | InChI=1/C11H9NO5S/c12-18(16,17)9-5-8(11(14)15)10(13)7-4-2-1-3-6(7)9/h1-5,13H,(H,14,15)(H2,12,16,17)/p-1 |
4-AMINOSULFONYL-1-HYDROXY-2-NAPHTHOIC ACID, 97 - Physico-chemical Properties
| Molecular Formula | C11H9NO5S |
| Molar Mass | 267.26 |
| Density | 1.4895 (rough estimate) |
| Melting Point | 225°C |
| Boling Point | 555.0±60.0 °C(Predicted) |
| Flash Point | 289.5°C |
| Vapor Presure | 3.74E-13mmHg at 25°C |
| pKa | 2.44±0.30(Predicted) |
| Refractive Index | 1.4790 (estimate) |
4-AMINOSULFONYL-1-HYDROXY-2-NAPHTHOIC ACID, 97 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S24/25 - Avoid contact with skin and eyes. |
4-AMINOSULFONYL-1-HYDROXY-2-NAPHTHOIC ACID, 97 - Introduction
Acid is an organic compound with the chemical formula C11H9NO5S. It has white or off-white crystals and is soluble in acids and alkalis. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: White or almost white crystal
-Melting point: about 140-145°C
-Solubility: soluble in acid and alkali, more difficult to dissolve in water and organic solvents
-Stability: relatively stable under normal conditions, but avoid exposure to strong light or high temperature
Use:
-Pharmaceuticals: acid is an important intermediate, widely used in the synthesis of drugs and biologically active compounds.
-Organic synthesis: It can be used as an important raw material in organic synthesis and participate in various important organic synthesis reactions, such as substitution reactions, condensation reactions, etc.
Preparation Method:
The preparation of the calcium acid is generally carried out by chemical synthesis methods. The specific method can be:
1.2-naphthoic acid is reacted with sulfuryl chloride to obtain 2-naphthoyl sulfuryl chloride.
2. react 2-naphthoylthionyl chloride with ammonia or amines to generate acid.
Safety Information:
Acid is generally considered to have low toxicity under normal use conditions and has little impact on the environment. However, as a chemical, it is still necessary to comply with safe operating procedures:
-Avoid contact with skin and eyes. If contact occurs, rinse immediately with water.
-Avoid inhaling aerosols, dust or vapors and operate with proper ventilation.
-Avoid contact with oxidants and acids during storage.
-Wear appropriate protective gloves and glasses during use.
The above information is for reference only. Please refer to the relevant literature and instructions for specific operation and use, and strictly abide by the laboratory safety operation procedures.
Nature:
-Appearance: White or almost white crystal
-Melting point: about 140-145°C
-Solubility: soluble in acid and alkali, more difficult to dissolve in water and organic solvents
-Stability: relatively stable under normal conditions, but avoid exposure to strong light or high temperature
Use:
-Pharmaceuticals: acid is an important intermediate, widely used in the synthesis of drugs and biologically active compounds.
-Organic synthesis: It can be used as an important raw material in organic synthesis and participate in various important organic synthesis reactions, such as substitution reactions, condensation reactions, etc.
Preparation Method:
The preparation of the calcium acid is generally carried out by chemical synthesis methods. The specific method can be:
1.2-naphthoic acid is reacted with sulfuryl chloride to obtain 2-naphthoyl sulfuryl chloride.
2. react 2-naphthoylthionyl chloride with ammonia or amines to generate acid.
Safety Information:
Acid is generally considered to have low toxicity under normal use conditions and has little impact on the environment. However, as a chemical, it is still necessary to comply with safe operating procedures:
-Avoid contact with skin and eyes. If contact occurs, rinse immediately with water.
-Avoid inhaling aerosols, dust or vapors and operate with proper ventilation.
-Avoid contact with oxidants and acids during storage.
-Wear appropriate protective gloves and glasses during use.
The above information is for reference only. Please refer to the relevant literature and instructions for specific operation and use, and strictly abide by the laboratory safety operation procedures.
Last Update:2024-04-09 15:17:59
Supplier List
Product Name: 4-AMINOSULFONYL-1-HYDROXY-2-NAPHTHOIC ACID, 97 Request for quotation
CAS: 64415-15-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 64415-15-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 4-AMINOSULFONYL-1-HYDROXY-2-NAPHTHOIC ACID, 97 Request for quotation
CAS: 64415-15-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 64415-15-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History
